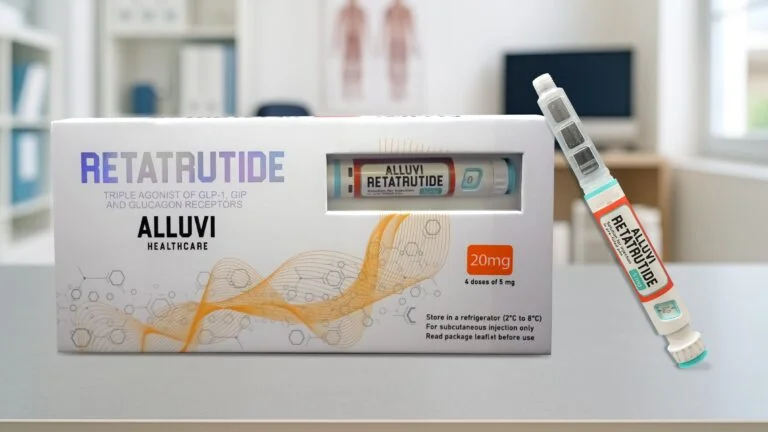
Alluvi Retatrutide 20mg
£115.00
Vls Retatrutide 20mg Pen, Alluvi Retatrutide 20mg Pen, Alluvi Retatrutide 20mg (R&D Only) is a research-grade formulation containing 20mg of Retatrutide, a novel investigational peptide.
Vls Retatrutide 20mg Pen | Alluvi Retatrutide 20mg Pen
Vls Retatrutide 20mg Pen, Alluvi Retatrutide 20mg Pen, Alluvi Retatrutide 20mg (R&D Only) is a research-grade formulation containing 20mg of Retatrutide, a novel investigational peptide. Retatrutide has attracted scientific interest for its potential role in metabolic regulation and weight management. This product is explicitly labeled “R&D Only”, meaning it is not intended for human or veterinary consumption and is not approved for clinical use. Its purpose is to support controlled laboratory studies, formulation testing, and mechanistic exploration.
Vls Retatrutide 20mg Pen | Alluvi Retatrutide 20mg Pen | Key Features
- Formulation strength: 20mg of Retatrutide per unit.
- Delivery system: Pre-filled research pen for precise and consistent dosing in experimental setups.
- Quality assurance: Sealed batches to maintain compound purity and stability.
- Storage requirements: Refrigeration at 2–8°C; freezing must be avoided.
- Shipping protocol: Cold packs included to preserve compound integrity during transit.
Vls Retatrutide 20mg Pen | Alluvi Retatrutide 20mg Pen | Research Applications
- Metabolic studies: Investigating Retatrutide’s potential effects on glucose regulation, insulin sensitivity, and energy balance.
- Obesity research: Exploring its role in appetite modulation and weight reduction in preclinical models.
- Formulation testing: Assessing stability, solubility, and compatibility with other compounds.
- Mechanistic exploration: Studying pathways related to peptide signaling, metabolic hormones, and cellular responses.
- Comparative analysis: Evaluating Retatrutide alongside other investigational peptides to understand synergistic or distinct effects.
Clinical Context
Retatrutide is currently under investigation in clinical research for its potential as a multi-receptor agonist targeting pathways involved in obesity and metabolic disorders. Early studies suggest it may influence weight loss and metabolic health, but Alluvi Retatrutide 20mg is not approved for therapeutic use. Its role remains confined to laboratory research, where scientists can explore its biochemical properties and potential applications. Translation into clinical practice requires extensive validation, peer-reviewed evidence, and regulatory approval.
Considerations
- Ethical compliance: Must be used strictly within research environments under institutional and regulatory guidelines.
- Safety protocols: Proper handling, storage, and disposal are essential to maintain compound integrity.
- Limitations: Laboratory findings cannot be extrapolated to clinical practice.
- Legal restrictions: Distribution and use are restricted to R&D contexts, not for consumption.
Be the first to review “Alluvi Retatrutide 20mg” Cancel reply
Related products
Alluvi Reta Pen
Alluvi Reta Pen
Alluvi Reta Pen
Alluvi Reta Pen
Alluvi Reta Pen
Alluvi Reta Pen
Alluvi Reta Pen
Alluvi Reta Pen






Reviews
There are no reviews yet.